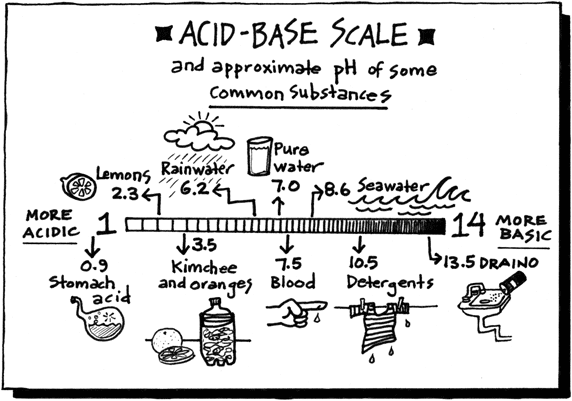
Acids, Bases, and the pH Scale
The terms acid and base describe chemical characteristics of many substances that we use daily. Acidic things taste sour. Basic or alkaline things taste soapy. Strong acids are corrosive and strong bases are caustic; both can cause severe skin damage that feels like a burn. However, mild acids and bases are common and relatively harmless to us. What makes a substance acidic or basic? The following equation is a good place to start:
We begin with two water molecules, and move some hydrogen atoms around. One water molecule gains a hydrogen and therefore takes on a positive charge, while the other water molecule loses a hydrogen atom and therefore becomes negatively charged. H3O+ is called a hydronium ion, and it makes things acidic. OH- is called a hydroxyl ion and it makes things basic. However, in water, there is a balance between hydroniums and hydroxyls so they cancel each others' charges. Pure water is neither acidic or basic; it is neutral.
So how does something become acidic or basic? That happens when the hydroniums and the hydroxyls are out of balance. If there are more positively charged hydroniums than negatively charged hydroxyls, then the substance is acidic. If there are more negatively charged hydroxyls than positively charged hydroniums, then the substance becomes basic. pH actually stands for the "potential (or power) of hydrogen."
Note: Sometimes people write H+ as a shorthand for H3O+ and that can cause confusion, because sometimes when people write H+ they really do mean only H+ and not H3O+. Watch out for that, and ask for clarification!
When we dissolve acids in water, we create an excess of hydroniums. When we dissolve bases in water, we create an excess of hydroxyls. Here are two examples. Vinegar, a weak acid, has a chemical formula of CH3COOH. When dissolved in water, it becomes CH3COO- and H+. The H+ ions combine with water molecules to form H3O+ so the solution becomes acidic. Now let's look at lye, a strong base with the chemical formula NaOH (sodium hydroxide). If we add NaOH to water, it dissociates into Na+ and OH-. The sodiums don't do anything important, but the hydroxyls make the solution more basic.
One last question: Why are strong acids and strong bases so nasty? It's because they are out of balance. They either have too many positive charges and are looking for negatives to get back into balance, or they have too many negative charges and are looking for positives to get back into balance. This makes them very reactive with anything they contact. When the positives and negatives are in equal number, they neutralize each other.