 Fernando P. Monroy
Fernando P. Monroy Fernando P. Monroy
Fernando P. MonroyImmunoparasitology: immunobiology of host-parasite
relationships
Ph.D., University of Queensland, Australia
Assistant Professor - Indiana State University (1993-1999)
Associate Professor - Indiana State University (1999-2000)
Joined the Department in 2000
Research conducted by Dr. Monroys
is directed towards clarification of the mechanisms employed by parasites
to survive and establish successful infections. More specifically, I am
interested in a group of parasitic organisms, the protozoa which includes
the agents of such terrible diseases as malaria, leishmaniasis, toxoplasmosis
and Chagas disease.
My research interest involves
the role of physiological stress in the pathogenesis of the obligated intracellular
parasite Toxoplasma gondii. Stress in its wider interpretation,
is a process central to the relationship between behavior and health. The
physiological responses to stress are mediated through neural and endocrine
systems. The two most likely routes by which the nervous system may communicate
with cells of the immune system is by release of catecholamines from the
sympathetic nervous system, and by secretion of glucocorticoid hormones
following stimulation of the hypothalamic-pituitary-adrenal axis. My research
goal intends to study the interactions between the nervous, endocrine and
immune systems by using an infectious agent as a marker. The obligated
intracellular parasite T. gondii is been used for two reasons: first,
to investigate the effects of stress as a cofactor in the pathogenesis
of an opportunistic infection in which cell-mediated immunity is of critical
importance in host defense. Second, T. gondii is the most common
opportunistic infectious agent of the central nervous system in HIV patients.
Our interests center on the effects of stress in the modulation of regulatory
cytokines [interferon (INF)-g , tumor necrosis
factor (TNF)-a , interleukin (IL)-12 and IL-10]
and neurohormones (glucocorticoids and catecholamine) in both the acute
and chronic phases of T. gondii infection.
LIFE CYCLE OF Toxoplasma gondii
 Secondly, I am interested in the role of catecholamines (Epinephrine and
nor-Epinephrine) as immune modulators in protozoan infection. My
current research centers in the initial interaction of the parasite with
the intestinal environment. During peroral infection with Toxoplasma
gondii, IFN-g is required
for survival in resistant strains (BALB/c); but in genetically susceptible
mice (C57BL/6) IFN-g contributes
to their early mortality. Mortality is associated with massive necrosis
of the villi and mucosal cells in the ilea ofC57BL/6
mice within 7 days. These histologic changesare mediated by
CD4+ T cells. It is clear that local regulation of IFN-g
following peroral infection is critical to determine whether this cytokine
is protective or detrimental and to determine genetic resistance/susceptibility
of the host to infection. Our laboratory evaluates the effects of
stress in an animal model of infections in which cell mediated immunity
is known to be of primary importance in host defense. Our studies
integrate the effects of a physical stressor, cold water stress (CWS);
manipulation of the the autonomic nervous system; T. gondii, a ubiquitous
protozoan parasite; and the natural route of infection (peroral).
Using this mild physical stressor my laboratory has studied its effects
in this host-parasite interaction during the acute phase after intraperitoneal
(i.p.) and peroral infection. CWS is detrimental to mice following
i.p. infection, but beneficial during peroral infection. We have
Secondly, I am interested in the role of catecholamines (Epinephrine and
nor-Epinephrine) as immune modulators in protozoan infection. My
current research centers in the initial interaction of the parasite with
the intestinal environment. During peroral infection with Toxoplasma
gondii, IFN-g is required
for survival in resistant strains (BALB/c); but in genetically susceptible
mice (C57BL/6) IFN-g contributes
to their early mortality. Mortality is associated with massive necrosis
of the villi and mucosal cells in the ilea ofC57BL/6
mice within 7 days. These histologic changesare mediated by
CD4+ T cells. It is clear that local regulation of IFN-g
following peroral infection is critical to determine whether this cytokine
is protective or detrimental and to determine genetic resistance/susceptibility
of the host to infection. Our laboratory evaluates the effects of
stress in an animal model of infections in which cell mediated immunity
is known to be of primary importance in host defense. Our studies
integrate the effects of a physical stressor, cold water stress (CWS);
manipulation of the the autonomic nervous system; T. gondii, a ubiquitous
protozoan parasite; and the natural route of infection (peroral).
Using this mild physical stressor my laboratory has studied its effects
in this host-parasite interaction during the acute phase after intraperitoneal
(i.p.) and peroral infection. CWS is detrimental to mice following
i.p. infection, but beneficial during peroral infection. We have
NEURO-ADRENO-IMMUNE
MACROPHAGES FROM STRESSED
INTERACTIONS
(above) AND CONTROL MICE.
 |
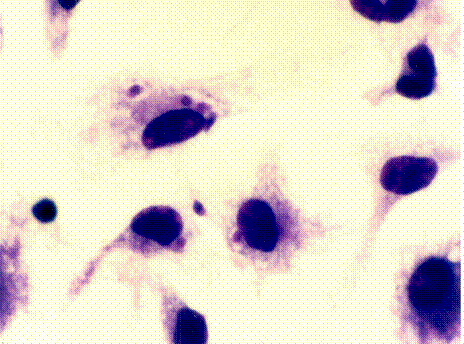
|
determined the effects of CWS on macrophage function (phagocytosis,
nitric oxide production, etc) and activation (calcium mobilization). Our
long-term
goal is to understand the contribution of stress or stress-induced
products to the outcome of infection in the intestinal environment. Intestines
are heavily innervated by sympathetic fibers and local secretion of neurotransmitters
(nor-epinephrine) is likely to affect the local environment and subsequent
immune responses. Our studies will investigate the contribution of
nor-epinephrine to the observed regulation of intraepithelial and lamina
propia lymphocytes during T. gondii infection.
Selected Publications:
MONROY, G.F. & ENRIQUEZ, F.J. (1992). Nematospiroides
dubius as a model for chronic intestinal parasitism.
Review Article for Parasitology
Today, 8(2): 14-17.
MONROY, G.F. & LOKER, E.S. (1993). Production of
heterogenous carbohydrate-binding proteins by the host snail
Biomphalaria glabrata following
exposure to Echinostoma paraensei and Schistosoma mansoni.Journal
of
Parasitology, 79: 416-421.
MONROY, G.F. & DRESDEN, M.H. (1996). Developmental
expression and role of cysteine proteinases in
Schistosoma mansoni.International
Journal for Parasitology, 25: 109-114.
MONROY, F.P. & DUSANIC, D.G. (1997). Mechanisms of
survival of Trypanosoma musculi in the kidneys of
chronically infected mice: kidney
form reproduction and immunological reactions. Journal of Parasitology,
83:848-852.
MONROY, F.P., MINNING, T. & DUSANIC, D.G. (1998).
Trypanosoma
musculi survival in the kidneys of
chronically infected mice: kidney
form ultrastructure , surface characteristics and serological interactions.
Journal
of Parasitology, 84:
914-919.
BANERJEE, S., AVILES, H., FOX, M.T. & MONROY, F.
(1999). Cold stress-induced modulation of cell immunity
during acute Toxoplasma gondii
infection in mice. Journal of Parasitology, 85: 123-132.
AVILES, H., BELLI, A., ARMIJOS, R., HARRIS, E. &
MONROY, F.P. (1999). PCR detection and identification of
Leishmania parasites in clinical
specimens in Ecuador: a comparison with classical diagnostic methods. Journal
of Parasitology, 85:
147-153.
MONROY, F., BANERJEE, S., DUONG, P. & AVILES, H.
(1999). Cold stress-induced modulation of inflammatory
responses and intra cerebral
cytokine mRNA expression in acute murine toxoplasmosis. Journal of Parasitology,
85: 878-896..
MONROY, F.P. & DUSANIC, D.G. (2000). The role of
sequestered parasites in chronic infections. Review article for
Parasitology Today, 16: 107-110.
AVILES, H. & MONROY, F. (2001). Immunomodulatory
effects of cold stress on mice infected intraperitoneally with
a lethal dose 50 (LD50) of Toxoplasma
gondii. NeuroImmunomodulation, 9: 6-13.
MORDUE, D.G., MONROY, F.P., LA REGINA, M., SCHREIBER,
R., DINARELLO, C. & SIBLEY, D. (2001). Acute
toxoplasmosis leads to overproduction
of Th1 cytokines. Journal of Immunology, 167:4574-4584.
AVILES, H. & MONROY, F. (2001). Toxoplasma
gondii: Cold stress-induced modulation of antibody responses.
Experimental Parasitology, 99:
89-96.
GETZ, J. & MONROY,
F. (2003). Effects
of ?-and ?-adrenergic
agonists on Toxoplasma gondii infection in murine
macrophages. Journal of Parasitology.
(in press).
AVILES, H., JOHNSON, M.T. & MONROY, F. (2003). Effects
of cold stress on spleen cell proliferation and cytokine
production during chronic Toxoplasma
gondii infection. NeuroImmunomodulation. (in press).